Background: Current standard of care for HR-MDS patients, i.e. hypomethylating agent (HMAs) treatment with azacytidine (AZA) or decitabine (DAC), is associated with a 30-50% response rate, and many efforts have been made to identify predictive factors for response. Fetal hemoglobin (HbF) levels have been proposed as a prognostic index for DAC treatment (Stomper J. et al, 2019), but no study has examined its prognostic value in AZA treated patients, and the mechanism of γ-globin chain re-expression has not been investigated.
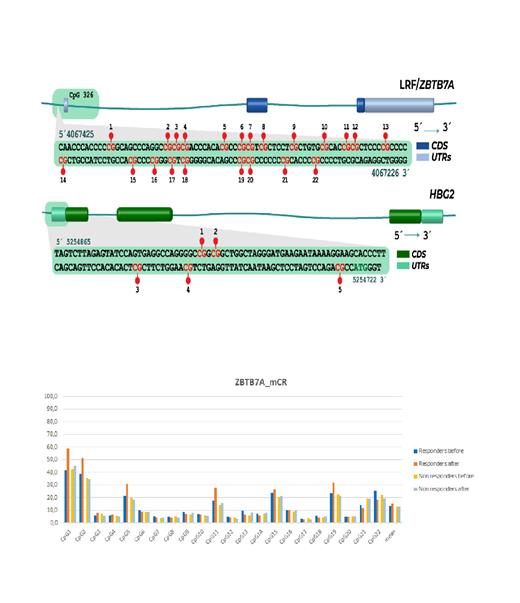
Methods: Bone marrow (BM) aspirates of 28 HR-MDS or CMML patients were collected at baseline and at the first evaluation of response, following 6 cycles of AZA treatment. Methylation of Cytosine followed by Guanosine (CpG) sites of the γ-globin gene promoter and CpG 326 of ZBTB7A/LRF, a previously characterized epigenetic repressor of HbF (Chondrou V. et al, 2022), were detected with pyrosequencing analysis in total genomic DNA, extracted from BM samples, pre- and post-treatment. Response to AZA was characterized as marrow response (mCR), when blasts were <5% and were reduced >50% from baseline, and as hematological response (HI-R) when there was at least one lineage improvement. HbF was measured with HPLC from the peripheral blood of 20 patients, at 5 time-points (before 0, 1, 2, 4 and 6-7 cycles of treatment), to test its expression pattern and potential prognostic significance. HbF values >1% were considered induced, as in previous studies (Stomper J. et al, 2019). For statistical analysis IBM SPSS 28.0.0.0 program was used. One patient with extremely high HbF before treatment (HbF 0=13.4%) was excluded from the analysis, as well as the CpG site 3 of γ-globin gene promoter, which was 100% methylated pre- and post- AZA treatment.
Results: Median baseline HbF levels were 0.6% (range 0 - 2.6), and they were increased to 0.8% (range 0 - 1.8) and to 1.1% (range 0 - 3.5), at 2 and 6 months, post-AZA treatment, respectively. Difference in the repeated measurements of HbF was of borderline statistical significance (p=0.068), specified between cycles 2 and 6 (p=0.038). Correlation analysis showed a significant association of mCR with induced HbF after 6 cycles of treatment (p=0.011, r s=0.618) and a strong positive correlation between elevation of HbF at cycle 2 and time to leukemia progression (p=0.019, r s=0.886). Overall, median methylation of the γ-globin gene promoter was 73.4% and 72.8% pre- and post-AZA treatment, respectively (p=0.159). However, patients exhibiting any kind of response showed reduced methylation at site 2 post- AZA, at the limit of statistical significance (p=0.095 for mCR patients and p=0.066 for HI-R), whereas site 4 was significantly hypermethylated among HI-R patients (p=0.042). A negative correlation between methylation of the γ-globin gene promoter and gene expression was also identified (p=0.008, r s=-0.427), particularly at the sites 2 and 5 (p 2=0.011, p 5=0.004).
Methylation analysis of CpG 326 of ZBTB7A/LRF showed significant hypermethylation at site 1 (p=0.044) and reduction of methylation at site 22 (p=0.043), following AZA treatment. When analysis was restricted among responders only, there was significant hypermethylation at site 1 (p 1=0.020) and borderline significant hypermethylation at sites 5 and 11, among mCR patients (p 5=0.069, p 11=0.079). Conversely, sites 9 and 13 were significantly hypermethylated among non-responders (p 9=0.034, p 13=0.047).
With Spearman's analysis, a significant negative correlation between methylation fold change in site 13 and mCR was observed (p=0.002, r s=-0.555). Although no correlation between HbF and CpG 326 methylation status pre- treatment was observed, after 6 cycles of AZA a borderline correlation was identified between HbF levels and fold change of mean methylation of CpG 326 (p=0.05, r s=0.482). Moreover, induced HbF levels were related at the limit of statistical significance to increase in methylation at sites 1,3,5,11 and 22 (p 1=0.077, p 3=0.052, p 5=0.087, p 11=0.061, p 22=0.060).
Conclusions: In this study, a relationship of γ-globin gene promoter methylation status and HbF levels was found among HR-MDS patients, treated with AZA. Moreover, hypermethylation in CpG326 of ZBTB7A/LRF after AZA treatment was associated with HbF levels, and different methylation patterns were observed between responders and non-responders. Further research is needed to clarify the role of LRF in MDS response to AZA.
Disclosures
Symeonidis:AbbVie: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Amgen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; BMS: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Gilead: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Janssen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Novartis: Consultancy, Honoraria, Research Funding; Pfizer: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Roche: Consultancy, Honoraria, Research Funding; Takeda: Consultancy, Honoraria, Research Funding, Speakers Bureau; Sanofi: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal